- [JOURNAL OF EXPERIMENTAL MEDICINE] Lymph node fibroblastic reticular cells regulate differentiation and function of CD4 T cells
- 관리자 |
- 2024-07-03 14:36:26|
- 142
- 2024-07-03 14:36:26|
[Title]
Lymph node fibroblastic reticular cells regulate differentiation and function of CD4 T cells via CD25
[Author]
Dongeon Kim, MingyoKim, Tae Woo Kim, Yong-ho Choe, Hae Sook Noh, Hyun Min Jeon, HyunSeok Kim, Youngeun Lee, Gayeong Hur, Kyung-Mi Lee, Kihyuk Shin, Sang-il Lee, and Seung-Hyo Lee
[Journal]
JOURNAL OF EXPERIMENTAL MEDICINE. 2022 Vol. 219 No. 5
[Abstract]
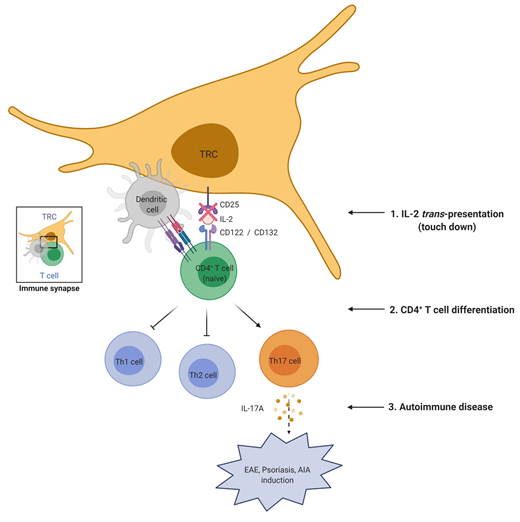
Lymph node fibroblastic reticular cells (LN-FRCs) provide functional structure to LNs and play important roles in interactions between T cells and antigen-presenting cells. However, the direct impact of LN-FRCs on naive CD4+ T cell differentiation has not been explored. Here, we show that T cell zone FRCs of LNs (LN-TRCs) express CD25, the α chain of the IL-2 receptor heterotrimer. Moreover, LN-TRCs trans-present IL-2 to naive CD4+ T cells through CD25, thereby facilitating early IL-2–mediated signaling. CD25-deficient LN-TRCs exhibit attenuated STAT5 phosphorylation in naive CD4+ T cells during T cell differentiation, promoting T helper 17 (Th17) cell differentiation and Th17 response-related gene expression. In experimental autoimmune disease models, disease severity was elevated in mice lacking CD25 in LN-TRCs. Therefore, our results suggest that CD25 expression on LN-TRCs regulates CD4+ T cell differentiation by modulating early IL-2 signaling of neighboring, naive CD4+ T cells, influencing the overall properties of immune responses.
Lymph node fibroblastic reticular cells regulate differentiation and function of CD4 T cells via CD25
[Author]
Dongeon Kim, MingyoKim, Tae Woo Kim, Yong-ho Choe, Hae Sook Noh, Hyun Min Jeon, HyunSeok Kim, Youngeun Lee, Gayeong Hur, Kyung-Mi Lee, Kihyuk Shin, Sang-il Lee, and Seung-Hyo Lee
[Journal]
JOURNAL OF EXPERIMENTAL MEDICINE. 2022 Vol. 219 No. 5
[Abstract]
Lymph node fibroblastic reticular cells (LN-FRCs) provide functional structure to LNs and play important roles in interactions between T cells and antigen-presenting cells. However, the direct impact of LN-FRCs on naive CD4+ T cell differentiation has not been explored. Here, we show that T cell zone FRCs of LNs (LN-TRCs) express CD25, the α chain of the IL-2 receptor heterotrimer. Moreover, LN-TRCs trans-present IL-2 to naive CD4+ T cells through CD25, thereby facilitating early IL-2–mediated signaling. CD25-deficient LN-TRCs exhibit attenuated STAT5 phosphorylation in naive CD4+ T cells during T cell differentiation, promoting T helper 17 (Th17) cell differentiation and Th17 response-related gene expression. In experimental autoimmune disease models, disease severity was elevated in mice lacking CD25 in LN-TRCs. Therefore, our results suggest that CD25 expression on LN-TRCs regulates CD4+ T cell differentiation by modulating early IL-2 signaling of neighboring, naive CD4+ T cells, influencing the overall properties of immune responses.





